
Several pharmacokinetic studies have been conducted in different ethnic groups of adults and showed different clearance (CL) for vancomycin both in Chinese patients ( He et al., 2014 Lin et al., 2015) and in Caucasian patients (4.03 ± 1.7 and 5.83 ± 2.39 L/h) ( Guay et al., 1993 Sánchez et al., 2010).

To obtain accurate estimation with this method for individualized therapy, it is crucial that reliable population pharmacokinetic characteristics are known for the target patients. The maximum a posteriori Bayesian estimation method has already been used to support vancomycin dosing decisions in adults ( Deng et al., 2013 Jacqz-Aigrain et al., 2015) and children ( Le et al., 2014).
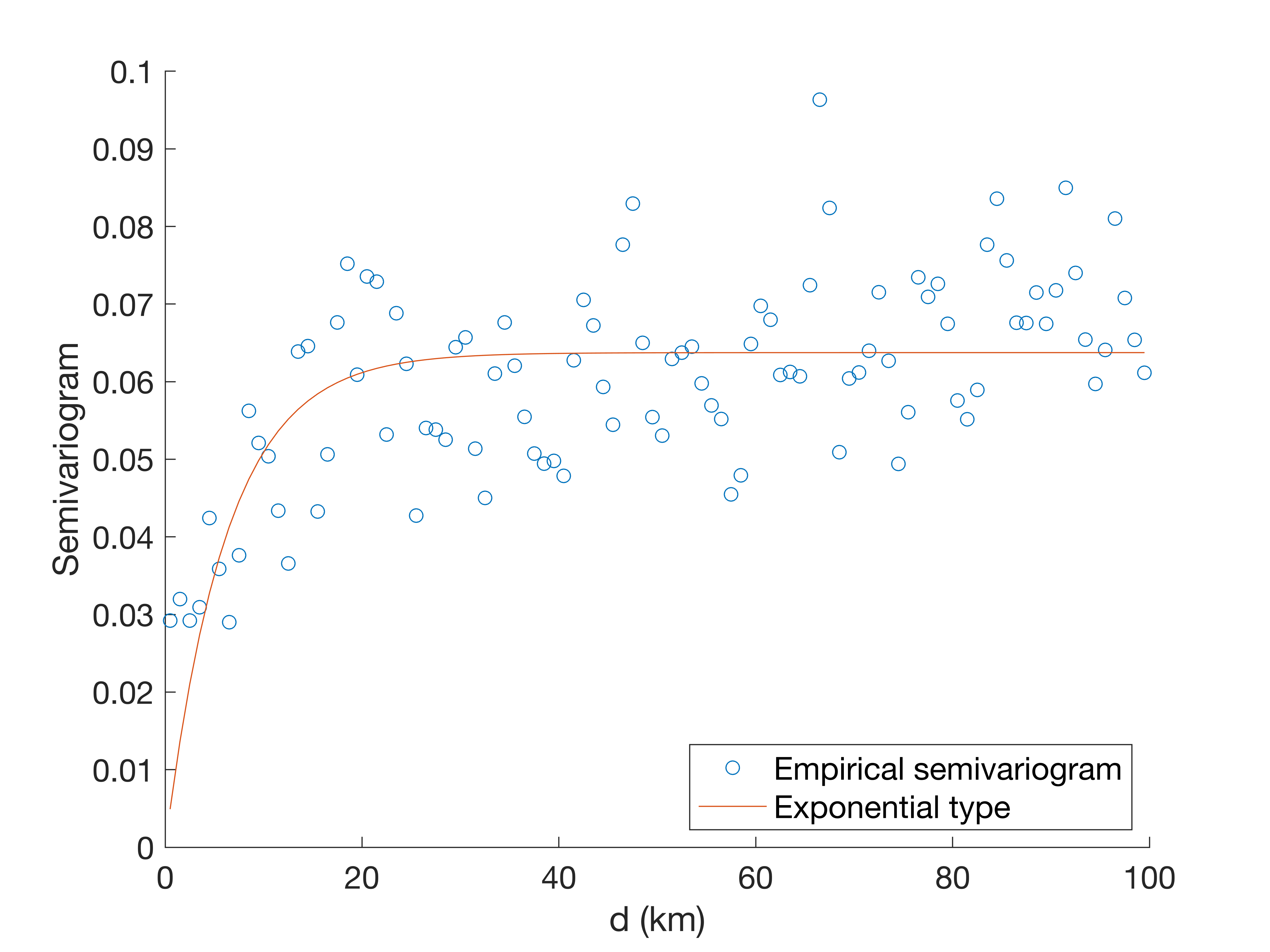
It is both challenging and imperative to optimize the vancomycin dosage regimen to achieve adequate exposure within a short period of time. In neonates, the pharmacokinetics of vancomycin are highly variable because of developmental and pathophysiological changes ( Stockmann et al., 2015). Vancomycin is mainly eliminated by the kidneys and is highly correlated with creatinine clearance. Low concentrations may result in less-effective therapy and an increased propensity for the development of bacterial resistance, whereas high concentrations are reported to be associated with nephrotoxicity and ototoxicity, although these toxic effects are less common in neonates ( Anderson et al., 2007). In 2011, guidelines issued by the Infectious Disease Society of America and pediatric-specific guidance recommended targeting trough concentrations of >15 mg/L for critically ill children and >10 mg/L for all other pediatric patients ( Liu et al., 2011). These findings will be valuable for developing individualized dosage regimens in the neonatal ICU setting.Īfter more than 60 years of widespread clinical use, vancomycin remains the gold standard antibiotic prescribed for the treatment of sepsis caused by coagulase-negative Staphylococci and methicillin-resistant Staphylococcus aureus in neonatal intensive care units (ICU) ( Tong et al., 2016). Recommended dose regimens for neonates with different Scr levels and postmenstrual ages were estimated based on Monte Carlo simulations and the established model. The exceptions to this are British National Formulary (2016–2017), Blue Book (2016) and Neofax (2017). Moreover, the established model indicated that in patients with a greater renal clearance status, especially Scr < 15 μmol/L, current guideline recommendations would likely not achieve therapeutic area under the concentration-time curve over 24 h/minimum inhibitory concentration (AUC 24h/MIC) ≥ 400. No obvious ethnic differences in the clearance of vancomycin were found relative to the earlier studies of Caucasian neonates. The average clearance was 0.309 L/h for a neonate with Scr of 23.3 μmol/L and body weight of 2.9 kg. Serum creatinine (Scr) and body weight were significant covariates on the clearance of vancomycin. The stability and predictive ability of the final model were evaluated based on diagnostic plots, normalized prediction distribution errors and the bootstrap method. Nonlinear mixed effect modeling was used to develop a population pharmacokinetic model for vancomycin. The study population consisted of 80 neonates in the neonatal intensive care unit (ICU) from which 165 trough and peak concentrations of vancomycin were obtained. The main goal of our study was to characterize the population pharmacokinetics of vancomycin in critically ill Chinese neonates to develop a pharmacokinetic model and investigate factors that have significant influences on the pharmacokinetics of vancomycin in this population. 6Department of Pharmacy, Gansu Provincial Hospital, Lanzhou, China.5Department of Pharmacy, Renmin Hospital of Wuhan University, Wuhan, China.4Department of Neonatology, Shanghai Children's Hospital, Shanghai Jiao Tong University, Shanghai, China.

